
Image: Jeff Fitlow / Rice University

Image: Ziyang Wang
Shengxi Huang and Ziyang Wang on mapping the molecular landscape of Alzheimer's disease without dyes, without tags, and without deciding in advance what to look for.
For most of its history, Alzheimer's research has proceeded through a necessary narrowing. Faced with a disease of staggering complexity, scientists identified specific targets, amyloid plaques and tau tangles, and built their methods around them. These tools were powerful, but they required a prior commitment: you chose your molecule, designed your marker, and saw exactly what you had aimed to see.
The SCOPE Lab approach begins with a different logic. Rather than focusing on a target, it asks a broader question: what is actually in this tissue, and where? By utilizing Raman spectroscopy, a technique that reads molecular fingerprints by how matter scatters light, the team observes brain tissue in its native chemical state. No dyes, no fluorescent tags, and no prior assumptions.
The resulting whole-brain molecular atlas was built from thousands of overlapping spectral measurements and analyzed by machine learning algorithms trained to find patterns invisible to human perception. It reveals a disease that is not confined to plaques nor spreads evenly. Instead, chemical changes are distributed in irregular, region-specific patterns. In the hippocampus and cortex, areas vital for memory, disruptions in cholesterol and glycogen metabolism appear alongside the expected accumulation of amyloid proteins.
Huang came to this work from optical spectroscopy and nanomaterials, not from biology. In her account, the brain slice shares fundamental traits with the 2D materials she has spent her career characterizing: it sits on a substrate, possesses a layered structure, and holds molecular information that light can be made to read. Ziyang Wang, who led the technical development, initially measured small areas before asking: what if we mapped the whole thing? What emerged was a map that challenges the window through which we have traditionally viewed this disease.
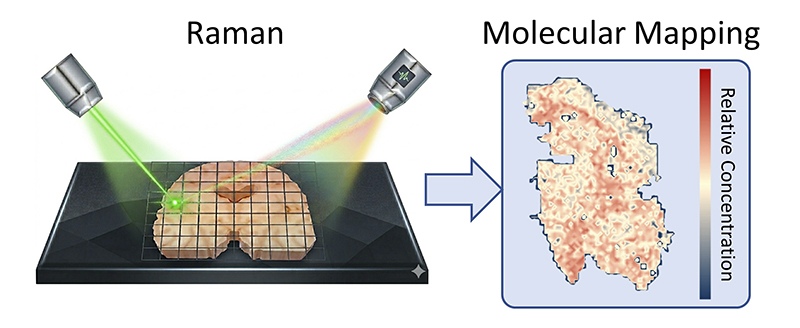
Image: Ziyang Wang / Shengxi Huang Research Group, Rice University
The Conversation
Shengxi Huang: My early training is in optical spectroscopy and nanomaterials. I also studied how 2D materials interact with organic molecules, a project that intrigued me because certain 2D materials can enhance the Raman signal of specific molecules. I began wondering if such an effect would work for more complex biomolecules and lead to practical applications. Toward the end of my PhD, I started working with biomolecules, and the results were exciting. After starting my own group, I decided to delve into the biosensing field using this unique phenomenon. This is the true gift of being a faculty member. You have the freedom to pursue ideas that may seem unusual. Fortunately, I met great collaborators who taught me about Alzheimer's and provided fantastic samples, which have led us to where we are.
Shengxi Huang: To us, a brain slice is like a piece of 2D material: it is flat, sits on a substrate, and carries Raman signals we can measure. What is more exciting about the brain is its versatility. There is a vast amount of molecular information to decipher across various conditions. From relatively simple 2D materials, we can already characterize defects, strain, thickness, and oxidation. With the much richer signals from the brain, we can learn far more. The data can become so complex that we need AI to understand certain signatures, a strong suit of electrical engineers. It is rewarding that these seemingly disparate fields have so much in common.
Ziyang Wang: Capturing the brain "as is" means observing its natural molecular state without introducing labels that may bias our observations. Traditional dyes and fluorescent tags require us to decide in advance which molecule to highlight, often focusing on known targets while overlooking the broader chemical environment. A label-free approach lets us measure intrinsic molecular fingerprints directly. Instead of looking only for predefined markers, we can observe the full molecular landscape and detect subtle biochemical changes that might otherwise remain hidden. This helps us understand the disease in a more holistic way.
Ziyang Wang: We wanted to take a broader view and ask how molecular changes are distributed across the entire brain. Advances in hyperspectral Raman imaging and AI made it possible to scan large sections and analyze the full molecular distribution. What surprised us most was that disease-related changes were not confined to a single region. The map revealed spatial patterns and regional progression, suggesting that Alzheimer's involves broader biochemical alterations than previously appreciated.
Ziyang Wang: Our findings suggest broader metabolic disruption rather than just the accumulation of misfolded proteins. While amyloid plaques and tau tangles remain hallmarks, we observed widespread changes in molecules such as cholesterol and glycogen. This indicates a systemic metabolic imbalance affecting energy use and lipid regulation in brain cells. Rather than viewing the disease only as localized protein aggregation, it may reflect deeper biochemical changes in how brain cells maintain metabolism.
Shengxi Huang: In biomedicine, many widely used tools like MRI, mass spectrometry, X-ray imaging and CT were originally developed by physicists. The story of Raman is similar. These tools provide fresh perspectives to understand diseases. Interdisciplinary work is exciting because a fresh perspective can sometimes lead to discoveries that those embedded within a field might not easily reach.
Ziyang Wang: Machine learning helps us detect patterns in extremely complex data, but it does not replace scientific reasoning. The algorithm highlights subtle molecular signatures that would be difficult for a human to recognize directly. However, the scientist remains essential for asking the right questions, designing experiments, and interpreting biological meaning. Human intuition guides hypotheses and ensures results make sense in the context of the disease. Machine learning is a powerful tool for discovery, while human insight remains central to validation.
Shengxi Huang: Yes, light becomes a tool to reveal hidden biochemical changes. While much of the work is technical, collecting spectra and analyzing data, stepping back to reflect on what these tools allow us to see can be inspiring. It reminds us that the goal is to better understand these diseases and contribute to ways of detecting and treating them earlier.
Shengxi Huang: Our lab focuses on sensing overall. The brain project is about sensing molecules associated with Alzheimer's, but our work with 2D and quantum materials also serves that purpose. By choosing the correct 2D materials to contact biological samples, we can significantly enhance the measured signal. We perform atomic engineering of 2D materials to generate new properties that serve as sensors. This fundamental study could lead to new quantum sensing regimes that provide precision no classical technique could match.
Ziyang Wang: Researchers may shift from studying isolated biomarkers to investigating how molecular changes spread across the brain. Instead of asking only whether a specific protein is present, scientists could examine how multiple biochemical pathways, such as lipid metabolism, change spatially and evolve during disease progression. I hope this enables us to identify the earliest molecular changes before visible pathology appears and discover new pathways involved in neurodegeneration.
Conclusion
There is a particular kind of scientific modesty in Shengxi Huang's description: the insistence that a fresh perspective can sometimes see what long familiarity cannot. Her lab did not arrive with a theory to confirm, but with a technique and the patience to build a method capable of answering what molecules are present and where.
What they found is both a result and a provocation. Alzheimer's is not a disease that lives in one place, nor is it simply a story of protein aggregation. It is a chemical disruption spread across an entire organ, detectable in the metabolism of cholesterol and glycogen, in the very chemistry of where memory once lived. Ziyang Wang asked: what if we could see the whole brain? The answer came back stranger and more complex than a single protein story allows. That strangeness, carefully measured, is the beginning of something.

