How a protein released by exercising muscle may hold the key to protecting the aging brain — without touching a single amyloid plaque
The Conversation
Part I: The Paradox of Plaque and Persistence
Henriette van Praag The link between plaques and cognitive decline is not as firmly established as many think. Post-mortem studies show that there are people with plaques who remained cognitively intact (PMID: 35332316). So having plaques is not always synonymous with dementia in terms of a person’s functioning. While we did not see a reduction per se in the number or density of plaques in our mouse model, we have not yet looked at the plaque composition itself — whether they are very dense, or what their measure of toxicity is.
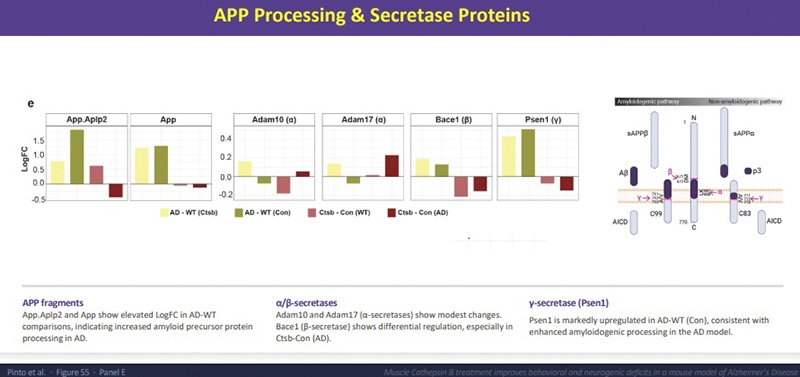
What I can say is that in our proteomics analysis, we do see modifications in the processing of amyloid at the molecular level. There are reductions in amyloid precursor protein and the enzymes that produce toxic fragments, like BACE1. Meanwhile, the non-amyloidogenic pathway enzymes ADAM10 and ADAM17 are elevated. We also see that Psen1, the gamma-secretase involved in producing the most toxic amyloid fragments, is markedly upregulated in untreated Alzheimer’s mice — a pattern that the treatment modifies. So it is not a total yes or no story — it is a modification of the amyloid environment, even if the plaques appear unchanged histologically.
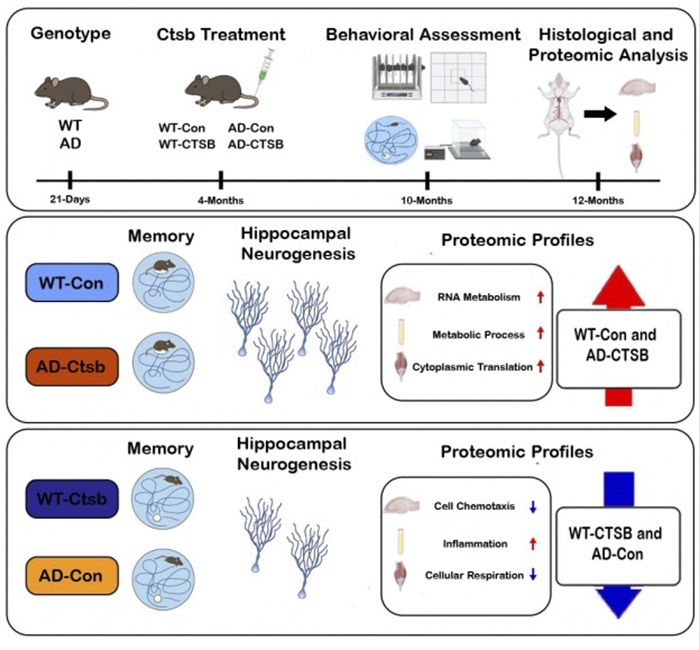
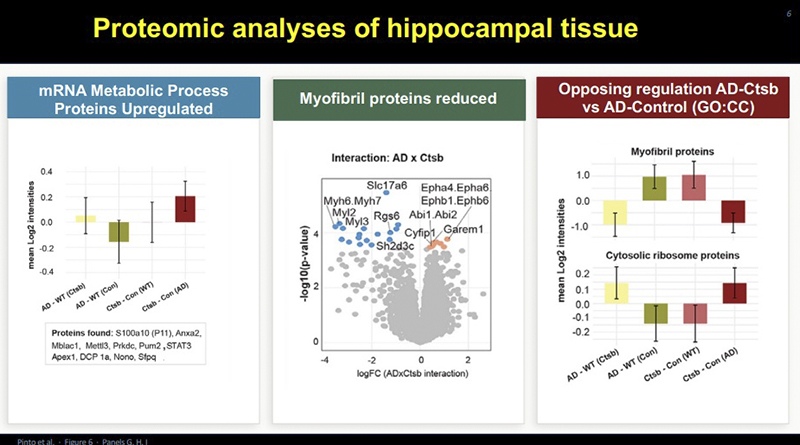
Henriette van Praag Several things may be happening. First, mRNA metabolic process proteins are upregulated resulting in modification of levels of proteins relevant to adult neurogenesis and synaptic plasticity, including those that mediate neural excitation and inhibition. In Alzheimer’s disease, there is often an over-excitability that leads to what we call excitotoxicity. Cathepsin B treatment reduces levels of glutamatergic proteins and increases GABAergic pathways — suggesting a rebalancing of the neural network from within.
Another observation is that in Alzheimer’s mice, we see an upregulation in myofibrillar proteins in the brain — proteins that have been reported to increase with aging and neurodegeneration in human brains. Our treatment reduces this presence, bringing the brain back toward healthy control levels. .
Part II: Molecular Mechanics and Evolutionary Context
Henriette van Praag I do not have a definitive answer, but the pattern we observe is striking. What happens in the muscle is very consistent with what happens in the brain. In the brain, we see increased RNA metabolism; in the muscle, we see improved cytoplasmic translation and metabolic processes. The brain is not doing one thing while the muscle does something completely different. They appear to work in concert, as one interconnected system.
Henriette van Praag This is a qualitative difference. The disease state apparently changes how the muscle processes this protein, and further research is needed to determine whether and how that affects everything downstream.
Part III: Therapeutic Translation
Henriette van Praag That is a possibility, a gene therapy to express Cathepsin B in skeletal muscle. It could be of interest to study muscle tissue from people with or without cognitive impairment to see how their cells respond to the vector in a laboratory setting. This could allow medical professionals to screen patients to determine whether the intervention might be relevant for them. Someone who already shows changes in muscle, and who cannot exercise, could potentially be identified and treated this way. However, we are still very far away from a safe and applicable intervention for people. Much more preclinical research is needed first.
Henriette van Praag That is exactly what I was thinking and what we discussed above. A biopsy or blood test could be a way of detecting early changes. Whether risk can be identified twenty years in advance remains unknown. But predicting risk from muscle health, before cognitive symptoms appear, is very much in line with where this research is pointing.
Part IV: Challenges and Limitations
Henriette van Praag Sarcopenia — the clinical loss of muscle mass that can come with ageing — is statistically linked to cognitive decline. Maintaining muscle health is not just about strength; it appears to matter for the brain as well. The general recommendation for older people is strength training, which helps not only mass but also metabolic activity and possibly the signaling signature that the muscle sends to the rest of the body.
Henriette van Praag Levels are the key. In certain cancers, concentrations of Cathepsin B in blood are much higher than what we see with exercise, and can be associated with poorer disease outcome.
Part V: The Future of Alzheimer’s Care
Henriette van Praag I very much agree that we should not treat the brain in isolation. A comprehensive approach — physical activity, healthy diet, improving metabolic health generally — would go a long way toward delaying or perhaps even preventing the onset of the disease. That is why I am talking to you: so you can make that recommendation to everybody, because I am just a scientist.
Henriette van Praag Oh yes, probably hundreds. Muscle is a vast endocrine organ. We have a long way to go to identify and study all of them, and especially the ones that are signaling to the brain.
Conclusion
Henriette van Praag’s work suggests that the brain does not age in isolation. It is in constant conversation with the muscle that moves it. When that conversation falters — when sedentary life silences the muscle’s molecular signals, when sarcopenia reduces the circulating myokines that maintain neuroplasticity — the brain loses a vital source of resilience it may not be able to replace on its own.
Cathepsin B does not clear the plaques. But it appears to rescue or restore, to some extent, what the plaques had disrupted: the brain’s capacity to generate new neurons, to balance its own excitation and inhibition, to maintain the protein landscape of a healthy mind. That is not a small thing. The question driving the next chapter of Alzheimer’s research may not be how to remove what has accumulated, but how to restore what has been lost.
The answer, it seems, may begin not in the neurology ward, but in the gymnasium.