
Columbia Scientists Transform Dairy Byproducts into Bioactive Injectable Hydrogels That Accelerate Tissue Repair
In a breakthrough that bridges food science and regenerative medicine, researchers at Columbia Engineering have developed a healing gel derived from an unlikely source: yogurt. This bioactive hydrogel represents a paradigm shift in tissue repair, harnessing naturally occurring particles called extracellular vesicles (EVs) from dairy products to create a material that not only delivers therapeutic molecules but also provides structural support for tissue regeneration.
Published July 25 in Matter, the study was led by Dr. Santiago Correa, assistant professor of biomedical engineering at Columbia Engineering. His team introduced a hydrogel system incorporating EVs extracted from milk, showing how everyday food byproducts can be transformed into advanced medical tools. Unlike synthetic hydrogels that often struggle to integrate with living tissue, this yogurt-derived gel mimics the body’s natural environment while actively promoting regeneration.
The breakthrough lies in the dual functionality of EVs, which act both as structural building blocks and as biological messengers that stimulate cellular communication and healing. In early mouse models, the injectable gel promoted new blood vessel growth and tissue repair within just one week—all without additional chemical additives. Its biocompatibility and ability to create an anti-inflammatory immune response suggest wide-ranging potential for wound healing and regenerative medicine.
Recently, we had the privilege of speaking with Santiago Correa about this groundbreaking work. The following is our conversation about the science, implications, and future of this novel approach to healing.
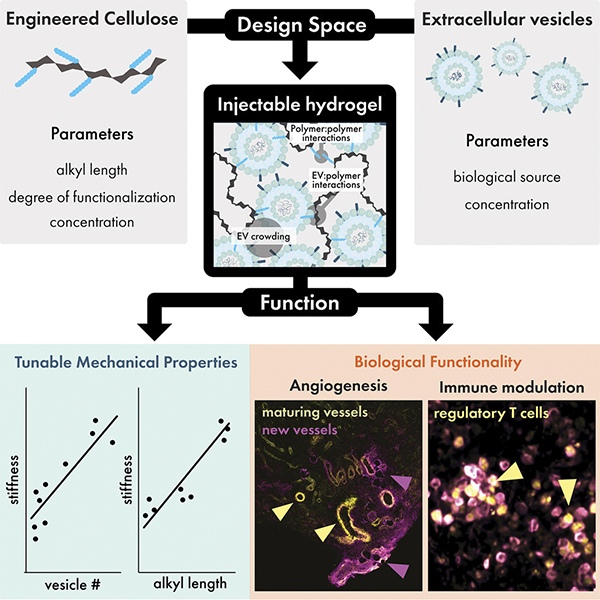
This interview has been edited for clarity and length.
Scientific and Technical Foundations
Your team used extracellular vesicles (EVs) from yogurt as both a structural and signaling component of the hydrogel. What surprised you most about their regenerative potential compared to more traditional mammalian-cell EVs?
We originally turned to yogurt EVs to solve a major bottleneck in developing the materials: EVs are notoriously difficult to obtain in large quantities. This has been a challenge in EV therapeutics for some time, but it was an even bigger hurdle for us because we were trying to make a material largely composed of EVs. When one of my students suggested pivoting to agriculturally sourced EVs—already gaining attention abroad but less so in the U.S.—we thought it could be a practical way to figure out the best way to build these hydrogels.
Deciding to test whether yogurt EVs had any biomedical potential was a bit of a risk. Many researchers might hesitate to use something that likely includes bacterial components from the yogurt-making process. I was curious, though, because probiotic EVs have been studied for their role in gut health and immune regulation. So we carried out a pilot study in mice. Honestly, I expected the immune system to react negatively, since these EVs came not only from another mammalian species but also from microbes. When my students reported visible blood vessel formation in the EV hydrogels, it was a stunning moment. It suggested that a regenerative program was taking place rather than rejection.
When you place this result in the broader context of EV therapies—which have mostly focused on mammalian EVs from highly specialized cells like stem cells—the yogurt EVs are not that different. Stem cell EVs, for example, are known to promote blood vessel growth and healing. The exciting part here is that if we can achieve similar effects using a byproduct of yogurt production, then we have a path toward affordable and scalable therapies without relying on complex human cell cultures.
One of the challenges in regenerative medicine is achieving both structural support and biological communication. How did the dual-role EVs help bridge that gap in a way synthetic materials often fail to do?
Many researchers have tried to incorporate EVs into materials, since EVs alone are quickly cleared from tissues or circulation. To be useful, they need to be concentrated and retained near the target site. Materials can help with that, but it’s been difficult to get EVs to stay put without chemically attaching them, which risks damaging them.
What’s different in our system is that EVs naturally “tie together” the polymers we use, forming crosslinks. We modify the polymers with fatty extensions, which dislike being in water. EV membranes have the same property: their lipid bilayer hides the water-repelling side. This creates a natural place for the polymers to attach. I often describe it as an ostrich burying its head in the sand—the fatty parts hide in the EV membranes. Enough of these interactions create a solid structure, but they’re also reversible. Under the pressure of a syringe, they break, and then reform after injection. That makes the hydrogel both stable and injectable.
Beyond structure, EVs bring their own biologically active cargo—proteins, lipids, sugars, and nucleic acids—that communicate with cells. By making EVs part of the hydrogel framework, we harness all of that complexity. Synthetic systems, by contrast, usually include only a handful of added signals, often fixed in place by chemical reactions. EVs, on the other hand, naturally present hundreds of signaling proteins on their surface. It’s a way of leveraging biology’s ability to create multifunctional nanomaterials—something we can’t replicate synthetically.
The tradeoff is that we don’t fully know which components are responsible for the observed effects. That’s the focus of our next studies: identifying the key features of these EVs that drive regeneration.
The gel mimics living tissue while stimulating new blood vessel growth. From a materials science perspective, how close are we to designing “living” biomaterials that not only repair but actively adapt to their host environment?
Living materials are a fascinating area of research. In my view, a truly living material would include engineered cells capable of sensing their environment and repairing or replacing the material as it degrades—much like how our bodies repair themselves after injury. What we have now are important steps toward that vision, but there’s still a long way to go.
Broader Implications for Medicine
Many regenerative treatments rely on chemical additives to trigger healing, yet your gel achieved results without them. Do you see this as a paradigm shift toward “chemical-free” biomaterials in future therapies?
I’d hesitate to call it “chemical-free,” since everything in biology is chemistry. Proteins, lipids, sugars, nucleic acids—these are all sophisticated biochemicals, and EVs are packed with them. The difference is that we didn’t need to manufacture or add them externally.
That brings two major advantages. First, cost: producing growth factors or engineered proteins is extremely expensive. Second, complexity: EVs carry far more signals than we could feasibly add one by one in a synthetic system. Of course, that means less control—but it also means leveraging millions of years of evolution, where cells have already refined highly effective signaling cocktails.
In your mouse model experiments, the immune system played a role in shaping tissue repair. Could this point to a future where immunoengineering and biomaterials are designed hand-in-hand for optimal healing outcomes?
Absolutely. This is one of the most exciting frontiers in the field. Researchers like Dr. Jennifer Elisseeff at Johns Hopkins have shown how immune cells respond to regenerative materials, identifying specific cell types that improve healing. These insights are now being adopted widely, giving us powerful tools to design biomaterials that actively guide the immune system toward better outcomes.
The fact that this material is injectable raises exciting possibilities for clinical use. How do you envision it being applied in real patients—wound healing, organ repair, or even more ambitious applications like heart or nerve regeneration?
We’re very excited about the possibilities, but it’s still early days. Injectability opens doors to many applications—from chronic wounds to organ repair—but we need to better understand the mechanisms driving these responses before choosing the right path. Personally, I’m especially interested in whether this approach could help address chronic wounds in diabetes, but time and science will guide us.
Accessibility and Sustainability
Using yogurt as a source for EVs is both scientifically clever and symbolically appealing. Do you see food-derived biotechnology becoming a major pathway for developing affordable, accessible medical treatments worldwide?
I think so. There’s growing interest in the therapeutic potential of probiotic-derived products. If they can deliver effects comparable to stem-cell EVs, then this could be a way to bring EV-based therapies much closer to real-world translation and affordability.
Future Outlook and Philosophy
This project brought together expertise from nanomaterials, polymer chemistry, and agricultural science. Do you think the future of biomaterials innovation lies less in disciplinary silos and more in cross-domain collaborations like this?
I strongly believe it does. The complexity of these challenges is simply too great for one discipline alone. In our lab, we emphasize teamwork, ensuring that expertise in materials, immunology, and clinical science all come together for each project.
Biomedical engineering often balances cutting-edge science with the deeply human goal of healing. How has this project shaped your own view of what it means to design technologies that not only repair the body but also restore quality of life?
For me, this project was eye-opening. Much of my past work focused on cancer and infectious disease—areas where the goal is to activate the immune system to fight. Here, we discovered regenerative potential instead, which led us to learn about diseases we hadn’t engaged with before, like chronic wounds and stroke. Hearing about the real struggles people face with these conditions has been a deeply motivating experience. It reminded me that our ultimate goal isn’t just advancing science, but improving lives.
Conclusion
This work with yogurt-derived EV hydrogels highlights how unexpected sources can inspire new directions in medicine. By combining affordability, accessibility, and regenerative potential, Santiago Correa’s team has opened the door to therapies that may one day reshape wound healing and tissue repair. Though still in early stages, the research illustrates the power of cross-disciplinary innovation—and reminds us that breakthroughs can come from even the most ordinary places.

